Individuals who inherit a variant of the TREM2 gene have up to a 4x increase in their risk for Alzheimer’s disease. A paper published in Nature Neuroscience with funding from Cure Alzheimer’s Fund offers insights into why individuals who carry this genetic mutation have an increased risk of Alzheimer’s disease and an accelerated rate of cognitive decline.
The research was led by three Cure Alzheimer’s Fund investigators including Dr. Jason Ulrich, Dr. Marco Colonna, and Research Strategy Council Member, Dr. David Holtzman. All three investigators are affiliated with the Knight Alzheimer’s Disease Research Center of Washington University School of Medicine, St. Louis.
The research used data from mouse models of Alzheimer’s disease as well as brain tissue from people who had been diagnosed with the disease to demonstrate that genetic mutations leading to decreased levels of TREM2 cause impairments in microglial function. Specifically, when microglia are unable to prevent amyloid-plaques from causing damage in the surrounding environment, tau spreads. When tau pathology spreads to areas of the brain such as the limbic system or neocortex, individuals are most likely to develop cognitive impairments.
Long before clinical symptoms of Alzheimer’s disease appear, two nefarious proteins accumulate in the brain: phosphorylated tau and amyloid-b. While amyloid-b may be first on the scene, tau is particularly damaging to the brain causing the cell death that is thought to trigger the memory impairments that characterize Alzheimer’s disease. A critical question that has puzzled the field has been how amyloid and tau are related. This research study indicates that impairments in the brain’s immune cells, or microglia, prevent amyloid from being reigned in. If the immune cells are unable to perform their tasks in the brain, the amyloid plaques damage the surrounding environment which accelerates the spread of tau tangles.
(BELOW IS REPRINTED FROM THE PRESS OFFICE OF WASHINGTON UNIVERSITY SCHOOL OF MEDICINE IN ST. LOUIS, TAMARA BHANDARI, JUNE 24, 2019)
In Alzheimer’s, amyloid clumps (blue) develop first in the brain, followed by tangles of the protein tau (red). Tau is associated with memory loss and confusion. People with a genetic variant that hobbles immune cells in their brains accumulate more tau near amyloid plaques than people with fully functional immune cells. Researchers at Washington University School of Medicine in St. Louis have found that immune cells that typically protect neurons from damage may be the link between such early and late brain changes in Alzheimer’s disease. Breaking that link could lead to new approaches to delay or prevent the disease.
Years before symptoms of Alzheimer’s disease appear, two kinds of damaging proteins silently collect in the brain: amyloid beta and tau. Clumps of amyloid accumulate first, but tau is particularly noxious. Wherever tangles of the tau protein appear, brain tissue dies, triggering the confusion and memory loss that are hallmarks of Alzheimer’s.
Now, researchers at Washington University School of Medicine in St. Louis have found that the link between the two proteins may lie in the brain’s immune cells that hem in clumps of amyloid. If the immune cells falter, amyloid clumps, or plaques, injure nearby neurons and create a toxic environment that accelerates the formation and spread of tau tangles, they report.
The findings, in mice and in people, are published June 24 in Nature Neuroscience. They suggest that reinforcing the activity of such immune cells – known as microglia – could slow or stop the proliferation of tau tangles, and potentially delay or prevent Alzheimer’s dementia.
“I think we’ve found a potential link between amyloid and tau that people have been looking for for a long time,” said senior author David Holtzman, MD, the Andrew B. and Gretchen P. Jones Professor and head of the Department of Neurology. “If you could break that link in people who have amyloid deposition but are still cognitively healthy, you might be able to stop disease progression before people develop problems with thinking and memory.”
While the formation of amyloid plaques and tau tangles have been recognized as key steps in the development of Alzheimer’s disease, researchers have struggled to pin down the relationship between the two. By themselves, amyloid plaques do not cause dementia. Many people over age 70 have some amyloid plaques in their brains, including some who are as mentally sharp as ever. But the presence of amyloid plaques seems to lead inexorably to the formation of tau tangles – the true villain of Alzheimer’s – and, until now, it wasn’t clear how amyloid drives tau pathology.
Holtzman and colleagues – including first authors Cheryl Leyns, PhD, a former graduate student in Holtzman’s lab, and Maud Gratuze, PhD, a postdoctoral researcher, as well as co-senior author Jason Ulrich, PhD, an assistant professor of neurology – suspected that microglia could be the link. A rare mutation in a gene called TREM2 leaves people with weak and ineffective microglia, and also increases their risk of developing Alzheimer’s by twofold to fourfold.
As part of the study, the researchers used mice prone to developing amyloid plaques and modified in various ways their TREM2 genes to influence the activity of their microglia. The result was four groups of mice: two with fully functional microglia because they carried the common variant of either the human or mouse TREM2 gene, and two with impaired microglia that carried the high-risk human TREM2 variant or no copy of the TREM2 gene at all.
Then, the researchers seeded the mice’s brains with small amounts of tau collected from Alzheimer’s patients. The human tau protein triggered the tau in mice to coalesce into tangle-like structures around the amyloid plaques.
In mice with weakened microglia, more tau tangle-like structures formed near the amyloid plaques than in mice with functional microglia. Further experiments showed that microglia normally form a cap over amyloid plaques that limits their toxicity to nearby neurons. When the microglia fail to do their job, neurons sustain more damage, creating an environment that fosters the formation of tau tangle-like lesions.
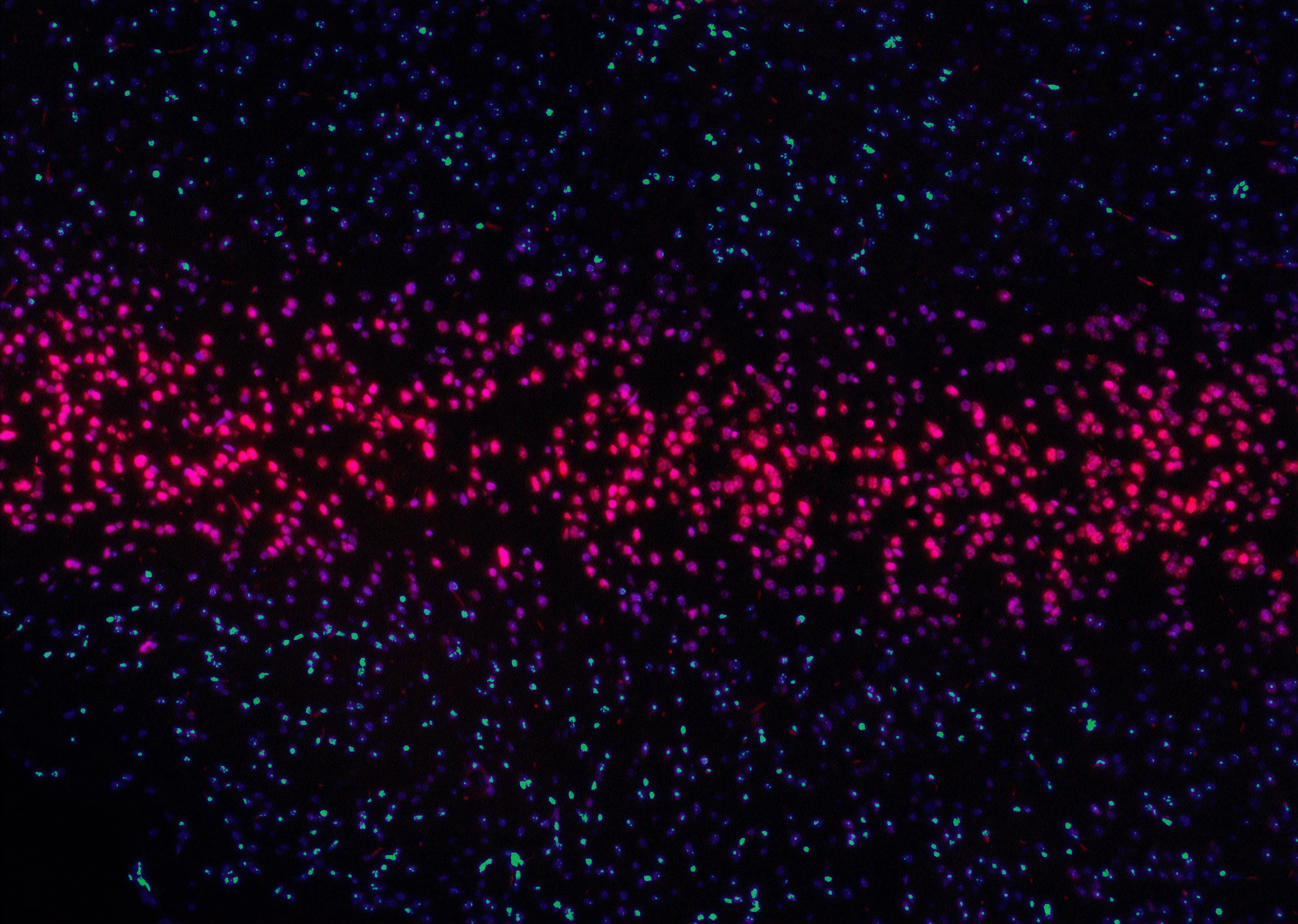
Amyloid protein (magenta) is visible in amyloid plaques (blue) surrounded by immune cells called microglia (green) and tau protein (red). Researchers at Washington University School of Medicine in St. Louis have discovered that faltering microglia may be the link between amyloid and tau – the two damaging proteins of Alzheimer’s disease. The findings could lead to a new approach for developing therapies for Alzheimer’s.
Further, the researchers also showed that people with TREM2 mutations who died with Alzheimer’s disease had more tau tangle-like structures near their amyloid plaques than people who died with Alzheimer’s but did not carry the mutation.
“Even though we were looking at the brains of people at the end of the Alzheimer’s process rather than the beginning, as in the mice, we saw the same kind of changes: more tau in the vicinity of amyloid plaques,” Holtzman said. “I’d speculate that in people with TREM2 mutations, tau accumulates and then spreads faster, and these patients develop problems with memory loss and thinking more quickly because they have more of those initial tau tangles.”
The converse also may be true, Holtzman said. Powering up microglia might slow the spread of tau tangles and forestall cognitive decline. Drugs that enhance the activity of microglia by activating TREM2 already are in the pipeline. It soon may be possible to identify using a simple blood test people with amyloid buildup but, as yet, no cognitive symptoms. For such people, drugs that break the link between amyloid and tau might have the potential to halt the disease in its tracks.
https://medicine.wustl.edu/news/alzheimers-missing-link-id-answering-what-tips-brains-decline/
The original research paper was published by Nature Neuroscience: “TREM2 function impedes tau seeding in neuritic plaques.” https://www.nature.com/articles/s41593-019-0433-0