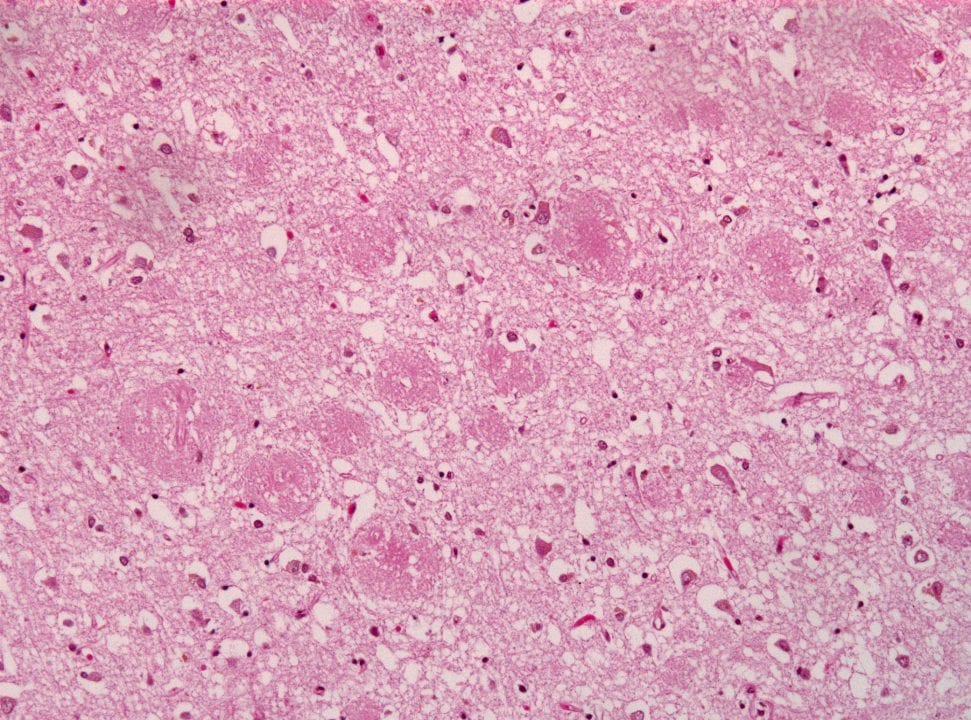
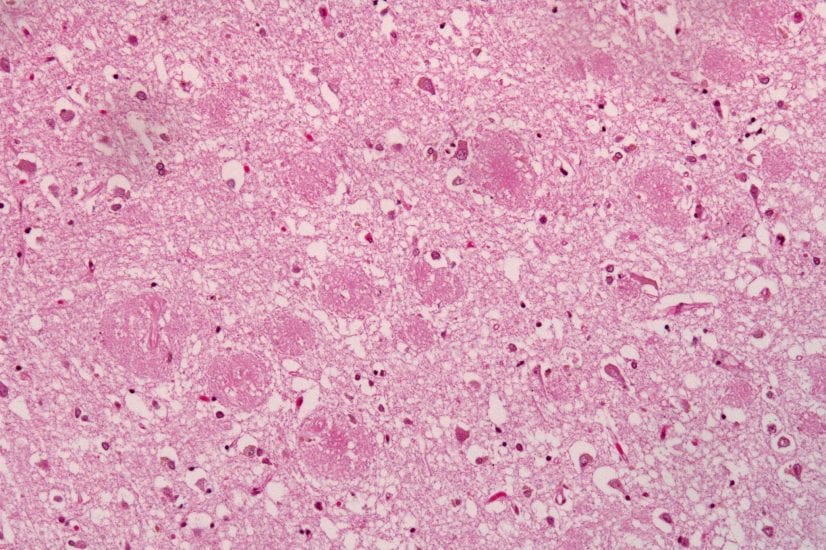
A new “alternative amyloid hypothesis” from the lab of Dr. Charles Glabe, at the University of California at Irvine, helps explain precisely how neurons (nerve cells) die in Alzheimer’s disease and how known genetic mutations initiate a chain reaction in this long process. The important new hypothesis was driven by research supported by Cure Alzheimer’s Fund, and has just been being published in the Journal Neurobiology of Disease. Dr. Glabe is a longtime member of Cure Alzheimer’s Fund’s Research Consortium.
The new approach is an “inside-out view” of Alzheimer’s, according to Glabe. The traditional view is that the protein fragment beta-amyloid aggregates into plaques outside neurons and subsequently causes stress and death to those neurons. Glabe’s new hypothesis proposes the reverse order: beta-amyloid forms first within the neuron, causing cell death, which subsequently spurs the formation of neuritic plaques. “It also has therapeutic implications,” he adds. “It suggests that gamma secretase modulators of the type that are being developed by Consortium member Dr. Steven Wagner will be successful because they will increase the secretion of soluble Abeta species and prevent the intraneuronal accumulation that leads to neuron death.” (Find out more about Dr. Wagner’s work here.)
“This represents an important challenge to our thinking,” says Dr. Rudy Tanzi, Chair of the Cure Alzheimer’s Fund Research Consortium. “The amyloid hypothesis has been strongly confirmed in recent years by our genetic and other research. But it’s important that we keep refining it, in order to continually improve intervention strategies.”
Read an abstract of the journal article here.